Precision Tumor Targeting Using Nanoparticles
Program-Specific Professor
Fuyuhiko Tamanoi
Fuyuhiko Tamanoi
Eradication of tumor growth is my research motivation. Cancer has been a major health problem for humankind throughout history. Siddhartha Mukherjee’s “The Emperor of All Maladies,” which won the 2011 Pulitzer Prize for General Non-Fiction, illustrates the 4000-year-old struggle against this relentless disease. I am determined to put an end to this long-lasting battle.

1977 Received PhD in Molecular Biology from Nagoya University
1977–1980 Postdoctoral fellow, Harvard Medical School
1980–1985 Principal Investigator, Cold Spring Harbor Laboratory, New York
1985–1993 Associate Professor, Chicago University
1994–Present Professor, University of California, Los Angeles
2017–Present Program-Specific Professor, iCeMS, Kyoto University
Shifting Focus From DNA Research to Cancer Therapy

I did not begin my career as a cancer researcher. I was a molecular biologist by education, and my earlier post-doctoral period was focused on DNA and RNA research. In the early 1980s, when I was at the Cold Spring Harbor Laboratory in New York, oncogenes (cancer-promoting genes) and tumor-suppressor genes were discovered, and these findings encouraged me to embark on cancer research, from my own perspective.
At that time, I was engaged in research on molecularly targeted cancer drugs. I researched anticancer agents that target the molecules that are expressed preferentially in cancer cells. These agents can attack aberrant cells more specifically than conventional cytotoxic agents, which also destroy normal cells, and my research was focused on the signaling pathways that regulate oncogenes. Research on molecular targets in cancer led to the discovery of trastuzumab (Herceptin), a ground-breaking therapy for breast cancer. However, my scientific instinct told me that targeting molecular signals would not be the ultimate solution for a cancer cure because cellular signaling systems are complex. For example, suppose that a particular oncogene signaling pathway is upregulated in certain cancer cell lines. Our logical approach is to attack that pathway. When we do, it upregulates a separate pathway. This sequence of attacking one pathway and activating another can go on and on.
Nanomaterials as a Therapeutic Tool
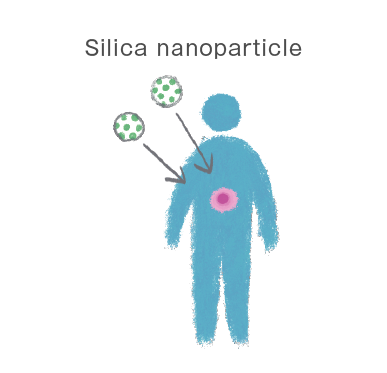
The above limitation with molecular target research led me to a totally different approach. Nanotechnology grabbed my attention. Nanomaterial carriers can be an effective tool to deliver anticancer agents to the target cells. For example, we employ mesoporous silica nanoparticles (ie, nano-sized particles with diameters 40–400 nm that contain thousands of pores). These particles are as small as virus particles, and can easily enter into cells. When nanoparticles that we developed are injected into the blood circulation, they do not leak out of healthy vessels. However, since cancerous tissues contain incomplete blood vessels, these particles can accumulate in the tumor as they leak out through the holes in vessel walls. This technique, which delivers the drug to the target site, is termed “the drug delivery system,” and we are using this method to cure cancer.
Mesoporous silica nanoparticles were first developed in 1988 by a group of researchers in Japan. One of their key features is stability. It generally takes 2 to 3 days before the injected nanoparticles accumulate in the tumor. This means that nanoparticle carriers must remain unchanged for a considerable time in the blood. Our silica nanoparticles meet this stability requirement. A possible disadvantage of a stable material is that it may accumulate in unintended or undesired areas of the body and thereby cause harm. To eliminate this undesirable possibility, we have recently created mesoporous silica nanoparticles that degrade after remaining for a sufficient amount of time in the human body. Another key feature is the porous structure. Our nanoparticles contain thousands of pores. The porous structure of a particle increases its surface area. Our mesoporous silica nanoparticles are estimated to have a total surface area of 100 ㎡ per gram. The particle’s highly hollow structure represented by this large surface area to weight ratio enables it to carry antitumor agents efficiently.
Porosity
Silica nanoparticles have countless pores, which means they have a large surface area to accumulate anticancer drugs. Each gram of silica nanoparticles has a surface area of 100㎡.
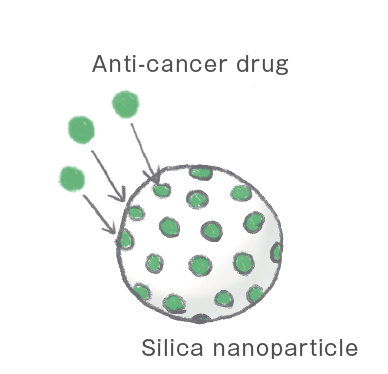
Stability
Silica nanoparticles can be placed in the body to deliver drugs to cancer cells. They have high stability and can exist in blood vessels for a while without breaking down.
Chick Egg Model to Test Anticancer Drugs
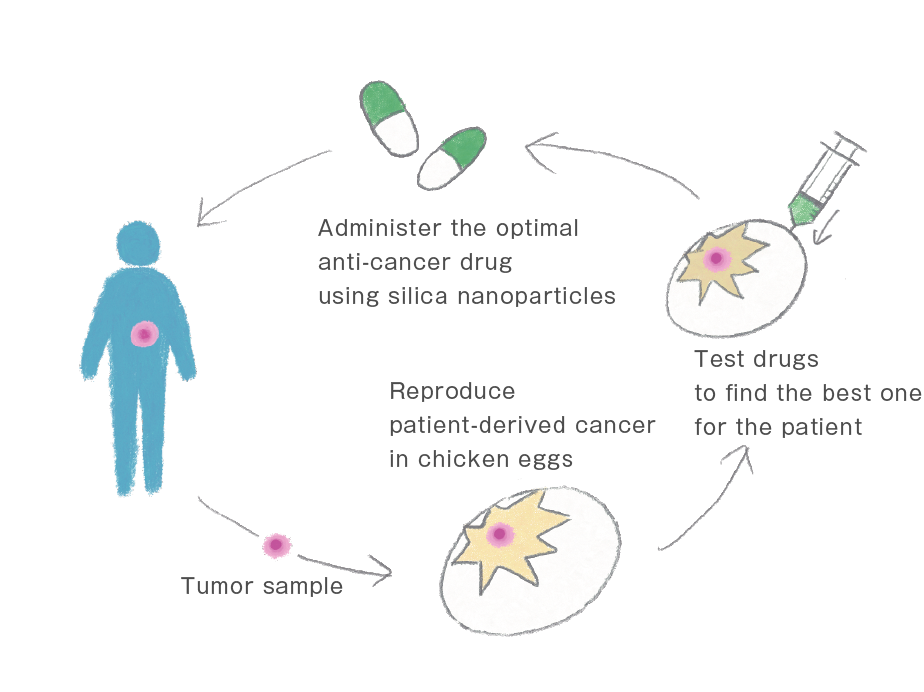
In addition to developing the silica nanoparticle-based drug delivery system, we are creating new methods to test the efficacy of anticancer agents. Specifically, we are using chicken eggs to develop a patient-derived xenograft model. I started to work on this method after I joined iCeMS, and we published our first report in 2018.
Many scientists use mice as a model to study cancer. However, creating a mouse tumor model is resource- and time-consuming, requiring several weeks before it can be used in experimentation. In contrast, chick egg models can be ready for use in a week. In this model, the chorioallantoic membrane (CAM) of a fertilized egg provides a scaffold for the growth of grafted cancerous tissue. Its production is simple and cost-effective. One fertilized chicken egg costs less than 100 yen (1 US dollar), whereas one laboratory mouse costs several thousand yen.
The CAM model can be prepared by using tumor samples harvested from patients. Moreover, it can be used to test and optimize the treatment for rare tumors for which large-scale clinical trials are difficult to conduct. Thus, the ease of creating the CAM model facilitates a wide range of applications. We have so far tested approximately one hundred different cancer cell lines and tumor samples, and approximately 70% of them were successfully cultured using the CAM model. Our preliminary results indicate that most of the major types of tumors can be established on the CAM membrane. We recently embarked on a joint project with the Japanese National Cancer Center to screen and identify the agents that act on a particular type of rare cancer.
Targeted Killing of Cancer Cells Using Radiology
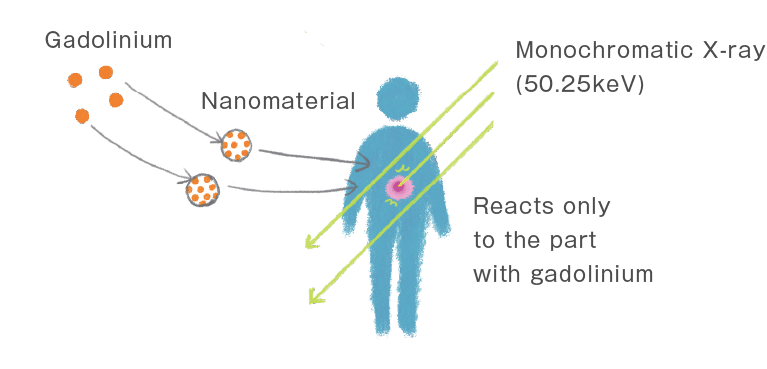
After the success of incorporating antitumor molecules onto the surface of silica nanoparticles, we began researching different treatment options. Our study using gadolinium started 3 years ago, and we developed a new radiotherapy in 2019.
Conventional radiation therapy irradiates beams made of a spectrum of energies, which have a harmful effect on healthy cells in the vicinity of the target tissue. By contrast, irradiation of monochromatic X-rays can minimize the adverse off-target effects. We produced gadolinium-loaded mesoporous silica nanoparticles. After these particles accumulated in tumor tissue, they were irradiated with special X-ray beams (50.25 keV). This caused gadolinium to emit electrons and destroy the tumor mass. This suggested the possibility of killing tumor cells in a targeted manner, without harming normal ones.
The use of monochromatic X-ray was chosen because we can take advantage of the Auger effect whereby energetic outer-shell electrons are emitted when high atomic number atoms, such as gold and gadolinium, are excited. The monochromatic X-rays of the defined energy level were generated by the SPring-8 synchrotron (Hyogo, Japan).
My Inner Drive Led Me to Become a Researcher
My father was a scholar of economics. His job required us to relocate frequently when I was a small child. I was born in Sendai, but grew up in Tokyo. At the age of 10, I spent one year in the USA because of my father’s work. It was a wonderful experience to be exposed to a totally different culture. I felt that America was the land of opportunity, and would give me opportunities to achieve my goals and dreams. After returning to Japan, I harbored the idea that I would go there again someday.

After becoming a student at the Department of Biophysics and Biochemistry, The University of Tokyo, I devoted myself to research. The laboratory that I joined was headed by Prof Fujio Egami. He was the founding director of the Mitsubishi-Kasei Institute of Life Sciences and one of the leading molecular biology pioneers in Japan. I was one of his last students. I was an undergraduate researcher when he retired.
Prof Reiji Okazaki was his successor. Prof Okazaki made several milestone achievements in molecular biology, including the discovery of “Okazaki fragments” during DNA replication, which were named in honor of him. After a while, Prof Okazaki returned to Nagoya University where he had been a faculty member. I also moved to Nagoya University and received a PhD degree.
At that time, graduate students were considered to be independent researchers, and the faculty expected them to think and act on their own. My colleagues and I had the ambition to develop our own research careers, and most of my fellow graduate students found their postdoctoral positions in the USA or Europe.
I found a position in the USA, and my childhood dream came true. Although most of my colleagues returned to Japan after several years of fellowship, I stayed overseas for more than 40 years. Looking back, I think that becoming a scientist was the natural course of my life.
Returning to Japan After Many Years in the USA
I first started to research DNA at the Harvard School of Medicine. I shifted my research interest to cancer after moving to the Cold Spring Harbor Laboratory in New York, as I explained earlier. While I was working on DNA there, the discovery of the first human proto-oncogene was reported. I was very impressed by this finding because it suggested that cancer can be studied using molecular biology techniques. I started to develop my own approach to cancer research.
After completing my term at Cold Spring Harbor, I moved to Chicago University, and then to the University of California, Los Angeles (UCLA). I am still affiliated with UCLA as a Professor and have been with the school for over 25 years.
At UCLA, people freely discuss various scientific topics. California’s mild weather and sunny skies may help people open up. People prefer going outdoors to staying inside. If you leave your workbench and go out for a walk, you come across someone you know. Consequently, you will learn a lot by strolling around the campus. Research institutions in the USA provide greater opportunities for discussing new ideas and thus provide excellent work environments.
I noticed many differences in the research atmosphere between Japan and the USA. One of the things that impressed me very much was that professors in the USA call and ask their students or students in other labs when they have something requiring clarification. Students also do not hesitate to ask questions to their professors and tutors. Some time ago, I visited Prof Bruce Alberts, one of the co-authors of “Molecular Biology of the Cell.” When I arrived at his office, he was in the midst of writing a textbook on molecular biology. When I greeted him, he said to me, “Give me a second. I have something I need to check.” He grabbed the phone, called one of the laboratory students, and started to talk about the matter he was concerned about. In America, the relationship between the faculty and students is smooth, interactive, and lively. Since I am accustomed to this type of relationship, I readily pick up my phone and call students and colleagues when something comes to my mind.

The way many American research institutions launch and conduct projects has both advantages and disadvantages. One of the disadvantages that I acknowledge is that people interact too much and creative and original ideas are less likely to arise. In fact, truly game-changing ideas are made and implemented more frequently in Japan than elsewhere. For example, mesoporous silica materials were first invented in 1988 by Prof Kazuyuki Kuroda and his colleagues at Waseda University. If Japan is a major seedbed for ground-breaking ideas, I should be in Japan to gain greater access to researchers with originality and creativity. This is the reason why I started to pay more attention to the research work in Japan.
I was deeply involved with iCeMS since its inception, having active discussions and relationships with its researchers. I took up a post at iCeMS in 2017. Kyoto University provides an ideal research environment with its excellent facilities for cancer research.
Innovative Cancer Therapy Can Change the Global Outlook

Cancer represents a serious threat to the human race. Many people lose their loved ones to cancer. Not only do patients with cancer suffer from its consequences, but their families do as well. Every time I hear about someone with cancer, I remind myself that I must do everything I can to leverage science and technology. The world may be a better place if a new treatment helps young people overcome cancer. I am determined to do whatever I can to combat cancer for the rest of my research career.
The carcinogenic process from normal tissue to malignant tumor has been elucidated for a number of cancer lines. For instance, the progression of colorectal cancer is associated with stage-specific changes in gene expression profiles. However, the knowledge of the pathogenetic mechanism does not immediately lead to a definite approach for a cure.
We are researching a drug screening method using the chick CAM model. Understanding the mechanisms of cancer development, progression, and metastasis is only the beginning. It has to be translated into viable therapeutic modalities. Our strategy will help identify effective anticancer agents, which must be investigated clinically. We need to thoroughly discuss the clinical efficacy and safety of a candidate cancer drug with medical experts. We have not yet come to this stage. My ultimate goal is to see patients with cancer cured by the treatment methods we develop. We are almost there.